|
The diagram that best shows the function of temperature is the correct answer. We want straight vertical lines in our graph because the phase transition is not changing because of the temperature. If we consider a solid going to a liquid at a point where all of the energy is being used to break bonds, the temperature stays the same. As for other state functions, the change in entropy for a process is the difference between its final ( Sf) and initial ( Si) values: S S f S i k ln W f k ln W i k ln W f W i For processes involving an increase in the number of microstates, Wf > Wi, the entropy of the system increases and S > 0. What is the difference between A and B? There is a gradual change at the phase transition point. 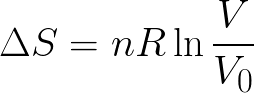
The more energy that a liquid has, the more it will have. 
Even if you have a solid that stays a solid and doesn't transition to a liquid as you add more temperature or energy particles, there will be more disorder than if you had a liquid. We assume that the entropy won't change as temperatures increase, like from solid to liquid to gas, and that's false. 
As temperature increases, we have flat lines. We want to know which one accurately depicts the change in entropy, we start with solid, liquid or gas. The graphs show the function of temperature. To calculate entropy changes for a chemical reaction We have seen that the energy given off (or absorbed) by a reaction, and monitored by noting the change in temperature of the surroundings, can be used to determine the enthalpy of a reaction (e.g.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
